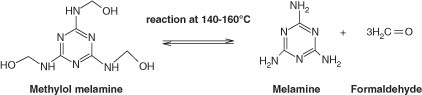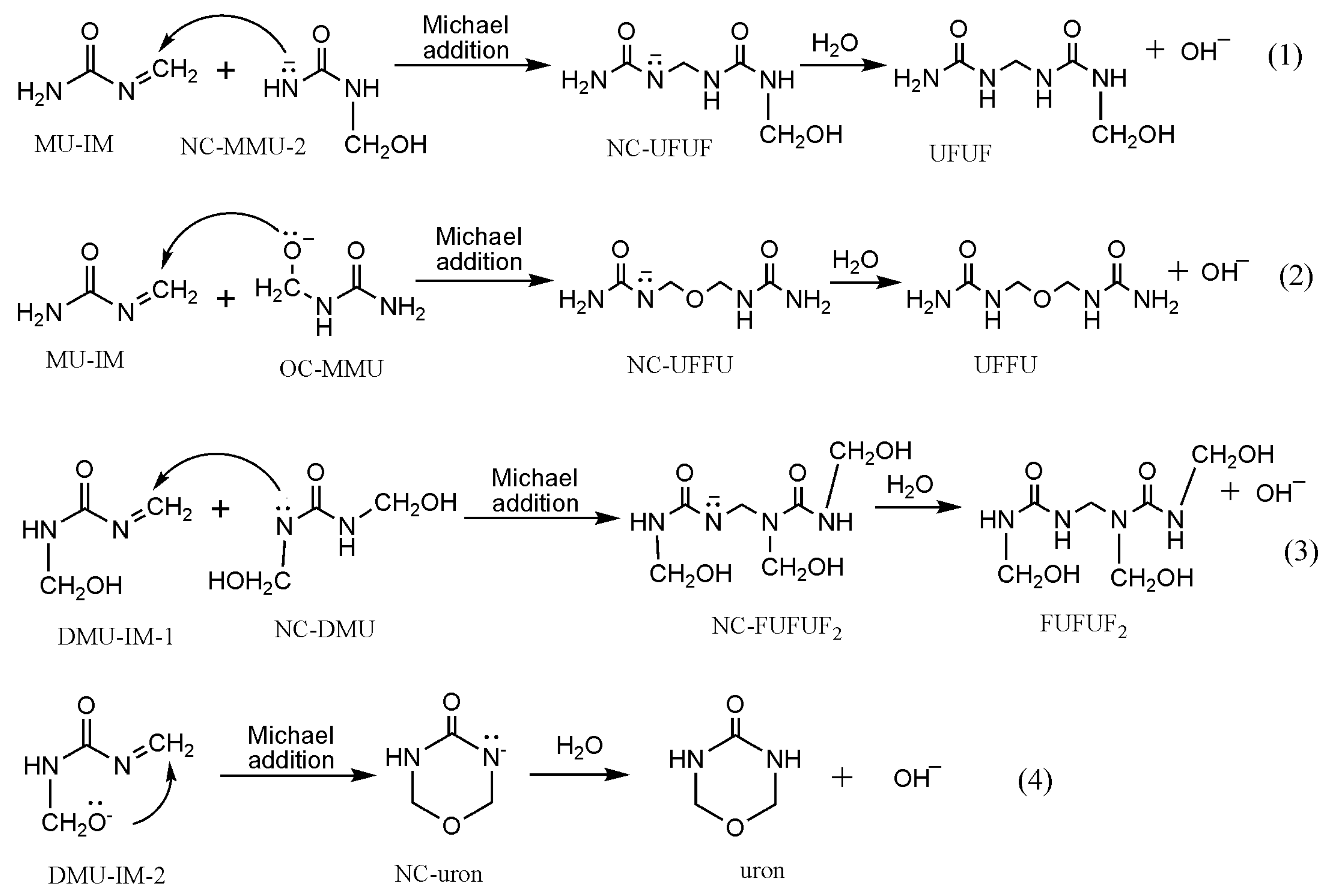
Polymers | Free Full-Text | New Mechanism Proposed for the Base-Catalyzed Urea–Formaldehyde Condensation Reactions: A Theoretical Study

Characterisation of melamine formaldehyde microspheres synthesised with prolonged microencapsulated reaction time

A review of recent progress in melamine-formaldehyde resin based nanocomposites as coating materials - ScienceDirect
Preparation of Formaldehyde-free Melamine Resin using Furfural as Condensation Agent and its Retanning Performances Investigat

Melamine–formaldehyde curing acceleration by TiO2-based silver-white pigments catalysis | SpringerLink
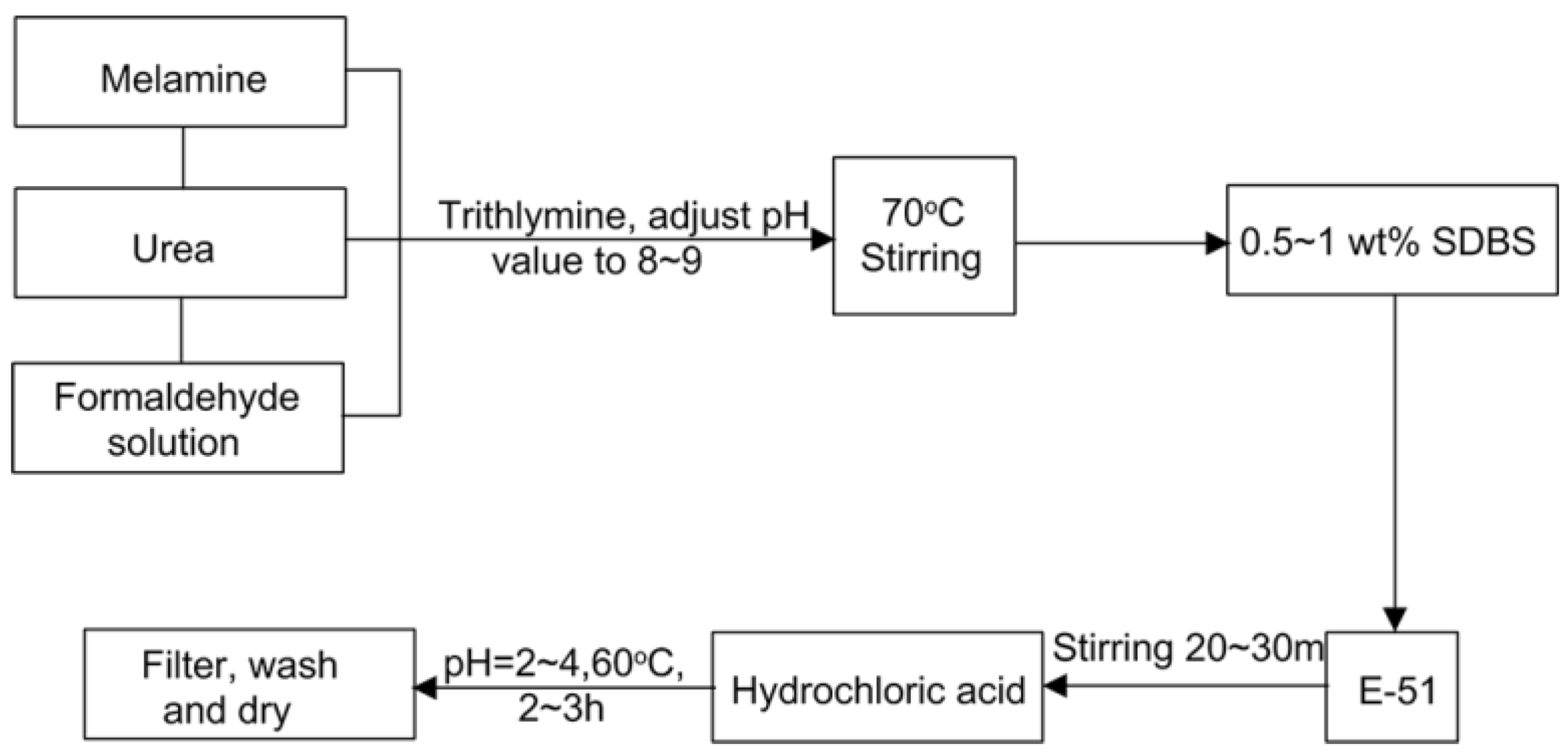
Materials | Free Full-Text | Preparation and Properties of Melamine Urea- Formaldehyde Microcapsules for Self-Healing of Cementitious Materials

Characterisation of melamine formaldehyde microspheres synthesised with prolonged microencapsulated reaction time

PDF) Using isoconversional kinetic analysis of liquid melamine-formaldehyde resin curing to predict laminate surface properties | Arunjunai Mahendran - Academia.edu

Hydrolysis of particleboard bonded with urea-formaldehyde resin for recycling. - Abstract - Europe PMC
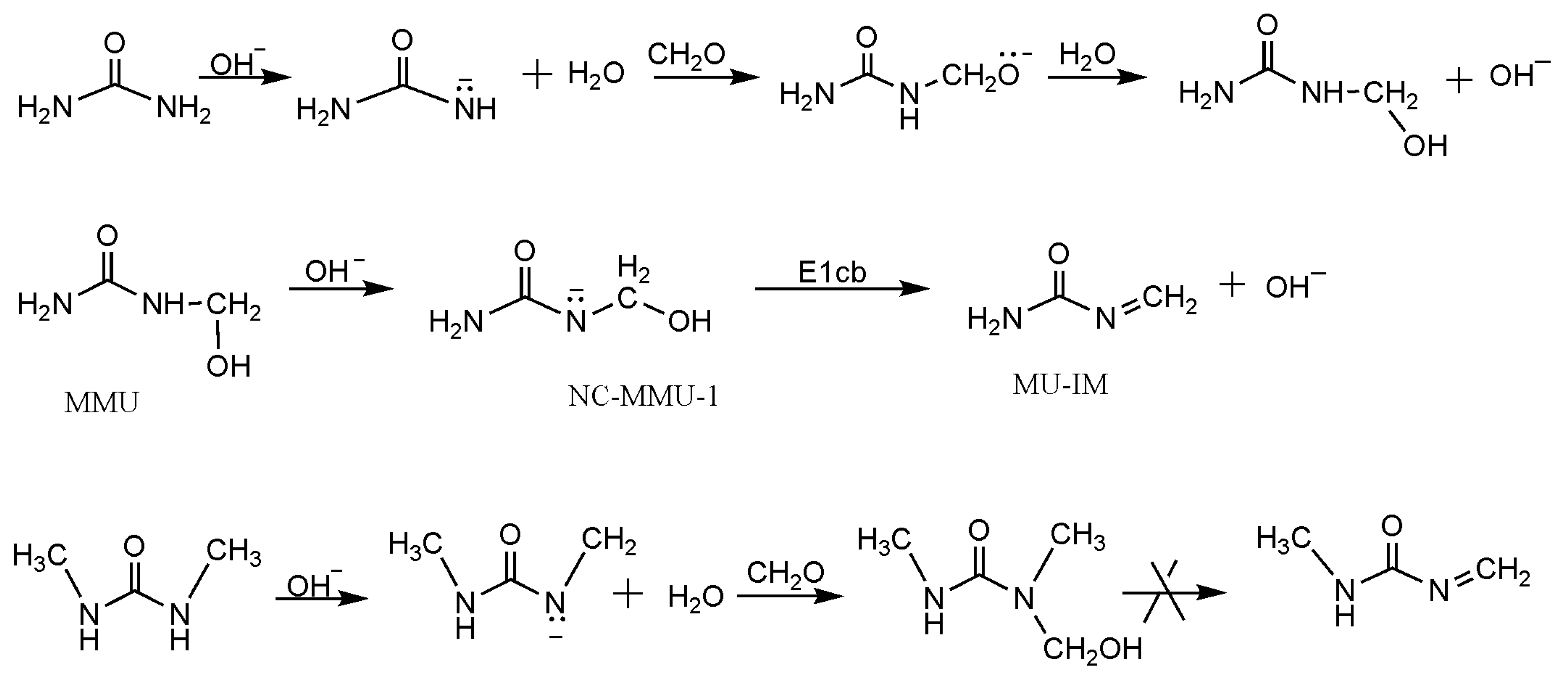
Polymers | Free Full-Text | New Mechanism Proposed for the Base-Catalyzed Urea–Formaldehyde Condensation Reactions: A Theoretical Study
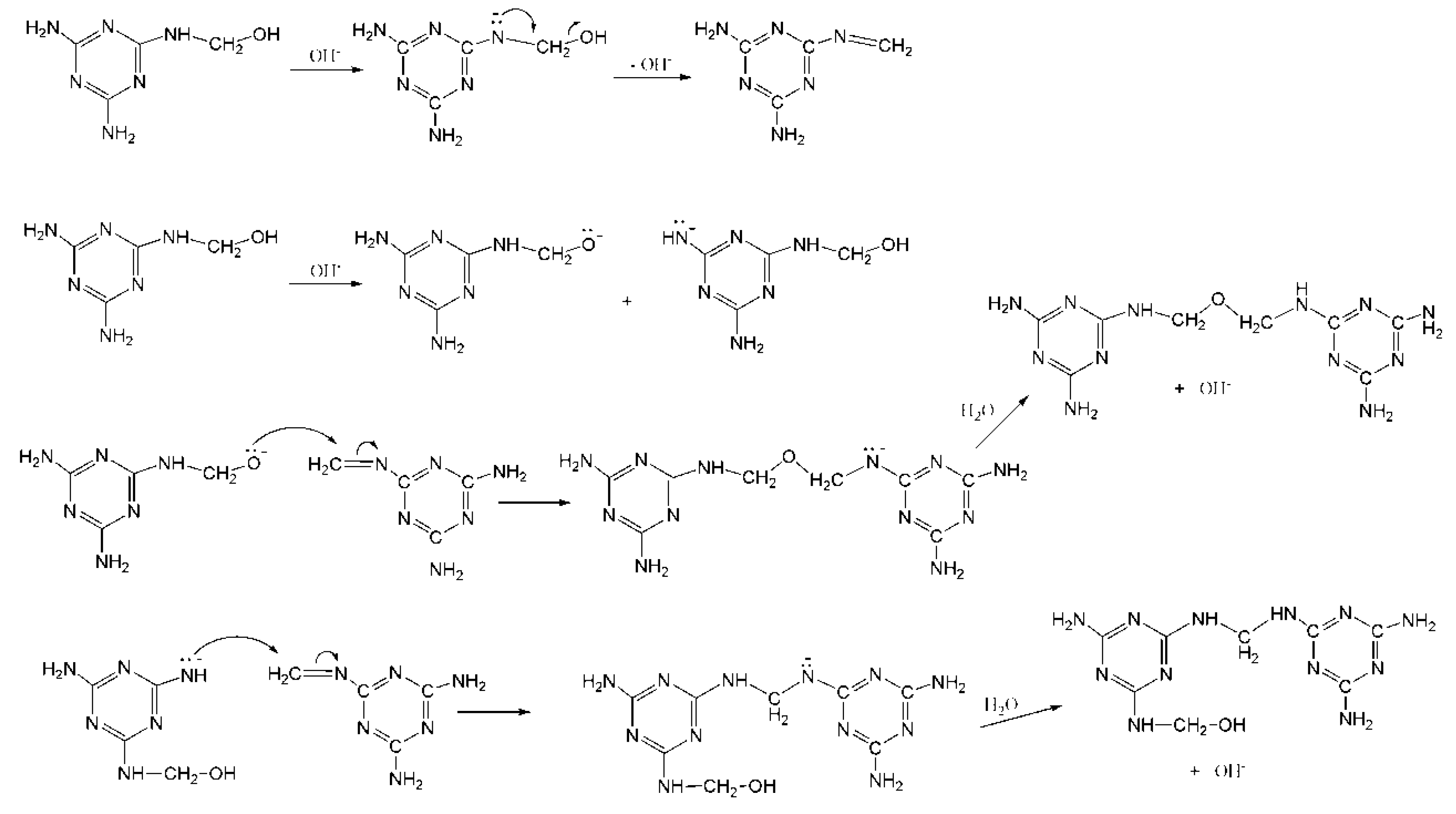



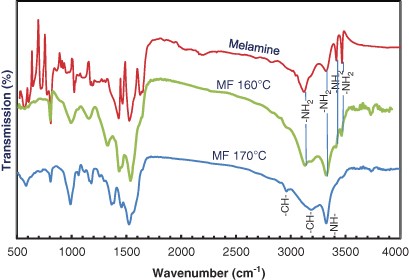

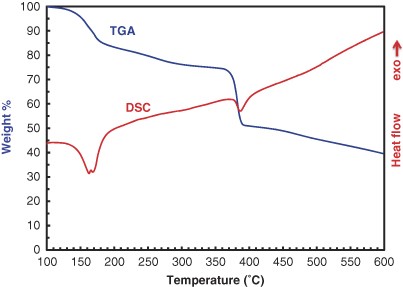

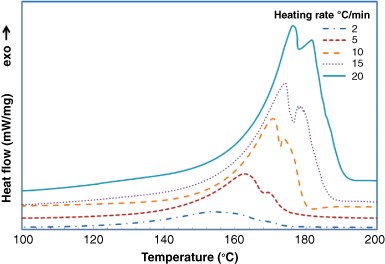
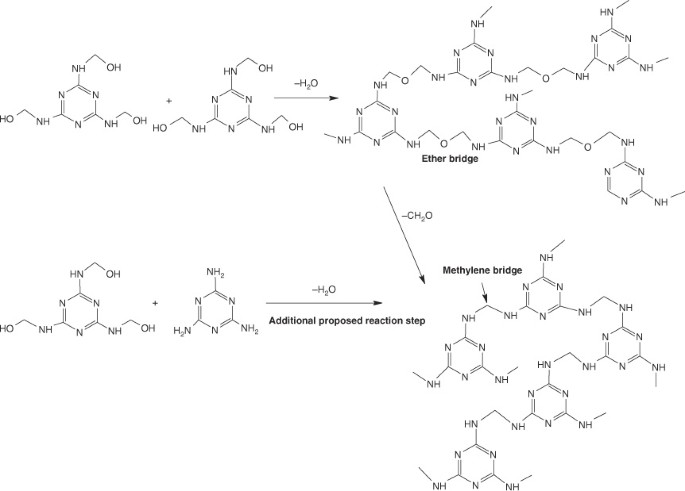
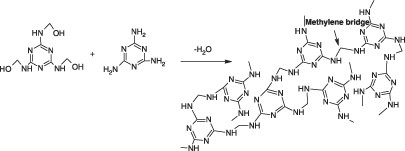
![PDF] Solid-State Microwave Synthesis of Melamine-Formaldehyde Resin | Semantic Scholar PDF] Solid-State Microwave Synthesis of Melamine-Formaldehyde Resin | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3fbce9dd2d7de7785d67f52b91119b6e91d296ed/4-Table1-1.png)