The Circle of Omicron Delta Kappa, Annual Report 2022, Vol. 101, No.1 by Associate Executive Director - Issuu

Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download

Real-fluid thermophysicalModels: An OpenFOAM-based library for reacting flow simulations at high pressure - ScienceDirect
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S

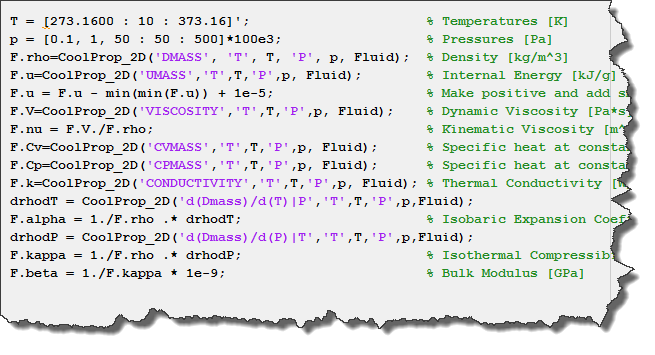
The equation is given below, CP=CV+TV(Beta2/K), links CP and CV with Beta and K. Use this equation to evaluate CP - CV for an ideal gas. CP=CV+T(Lambda P Lambda T)V(Lambda V Lambda

Cardiac toxicities in multiple myeloma: an updated and a deeper look into the effect of different medications and novel therapies | Blood Cancer Journal