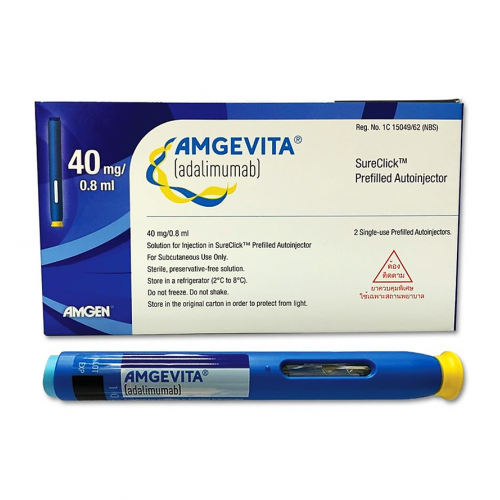
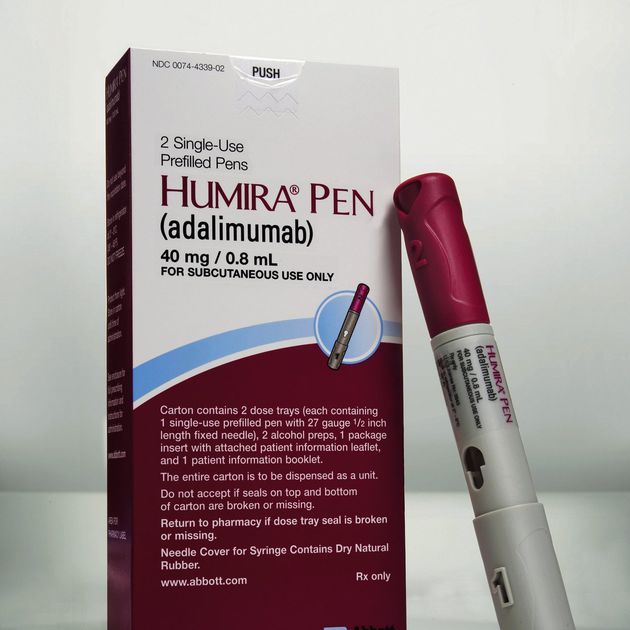
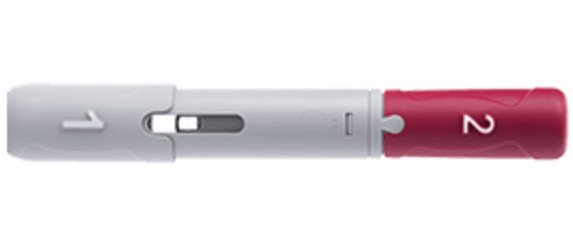
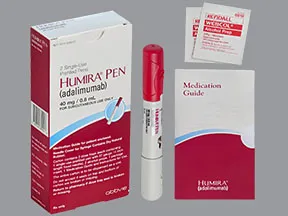
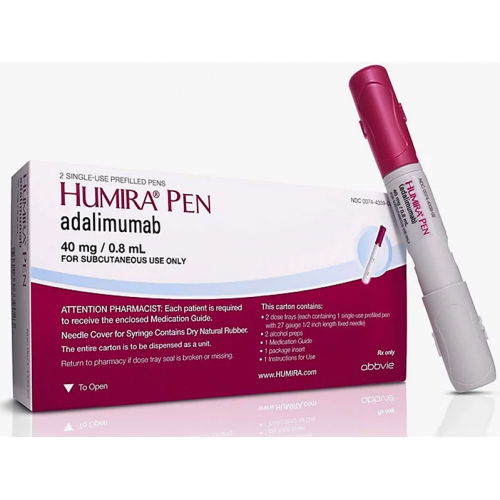
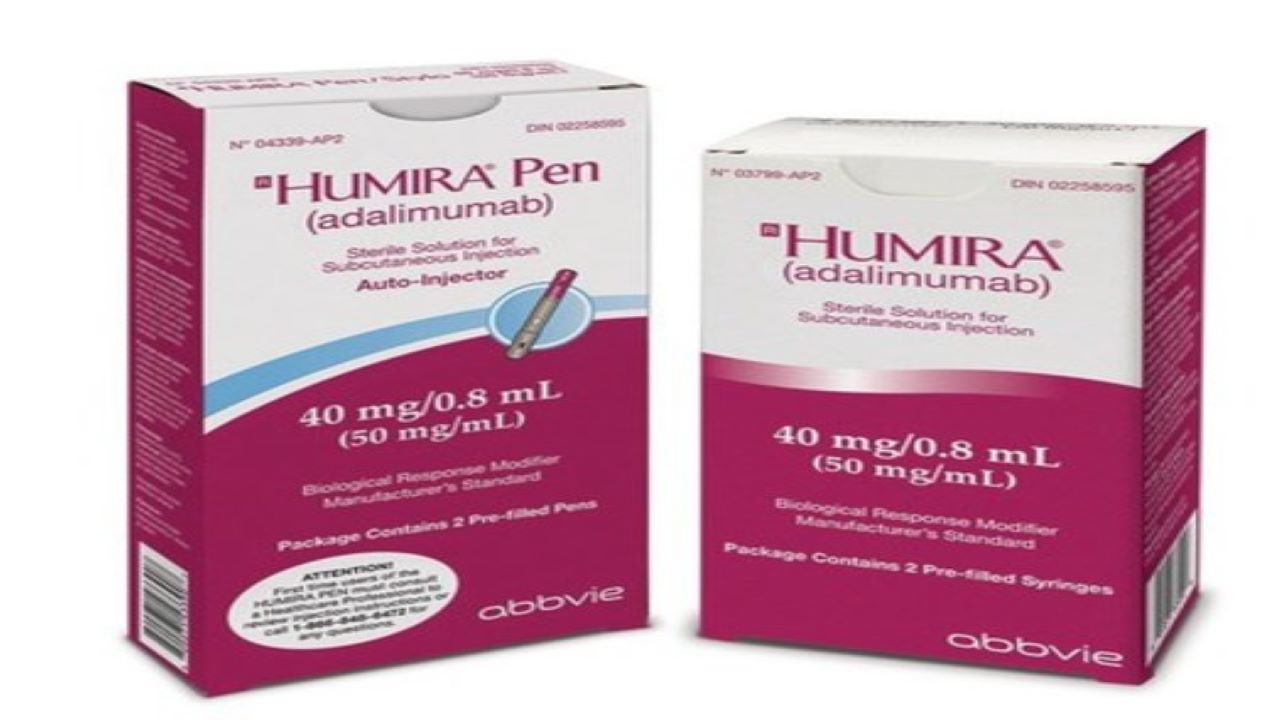
AbbVie and Eisai Announce the Launch of HUMIRA® Pen, an Auto-InjectorFormulation for Fully Human Anti-TNF-α Monoclonal Antibody HUMIRA® | Investors(2018) | EA Pharma Co., Ltd.
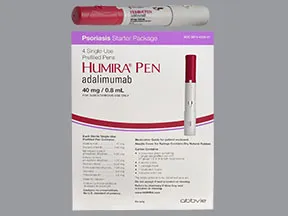
Humira Pen Psoriasis Starter Pack subcutaneous: Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD