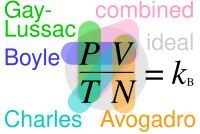
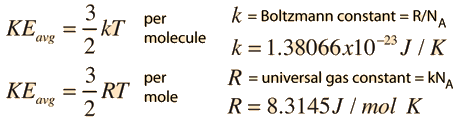SOLVED: Boltzmann constant: kB = 1.38 x 10-28 JK Thermal energy at 300 K: kBT = 0.0259 eV Energy unit conversion: 1eV = 1.60 x 10-19 J

Twitter 上的 MathType:"The Boltzmann Constant was introduced by Max Planck in the derivation of the law of black body radiation. It is used in thermodynamics and represents the proportionality factor between the
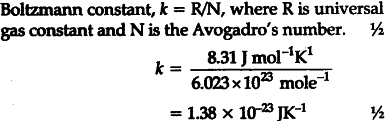
What is meant by Boltzmann constant ? Calculate its value in S.I. units - CBSE Class 11 Physics - Learn CBSE Forum

In the relation, P = alpha/betae^alphaz/ktheta P is pressure, Z is distance, K is Boltzmann constant and theta is the temperature. The dimensions of beta will be